Arbeitsgruppe Garbers
Cytokines in Inflammation and Cancer
Research Scope
The major focus of our group is to understand the role of of Interleukin (IL-) 6 family cytokines in development, inflammation and cancer.
IL-6 family cytokines are small, glycosylated proteins that can in principle be secreted by all cells of the human body. They exhibit pro- as well as anti-inflammatory properties and can act in a systemic or local fashion. IL-6 type cytokines are involved in a variety of physiological processes, ranging from differentiation and proliferation to apoptosis of their target cells.
Most cytokines bind to their target cells through a unique combination of membrane-bound α- and β-receptors. For instance, IL-11 binds in a first step to the IL-11Rα, and this complex subsequently recruits two gp130 β-receptors, which are the signal-transducing subunits of the IL-11 signaling complex. The Janus kinase (Jak) / Signal Transducer and Activator of Transcription (STAT) pathway is the major intracellular signaling pathway activated, leading to transcription of specific target genes (Figure 1).
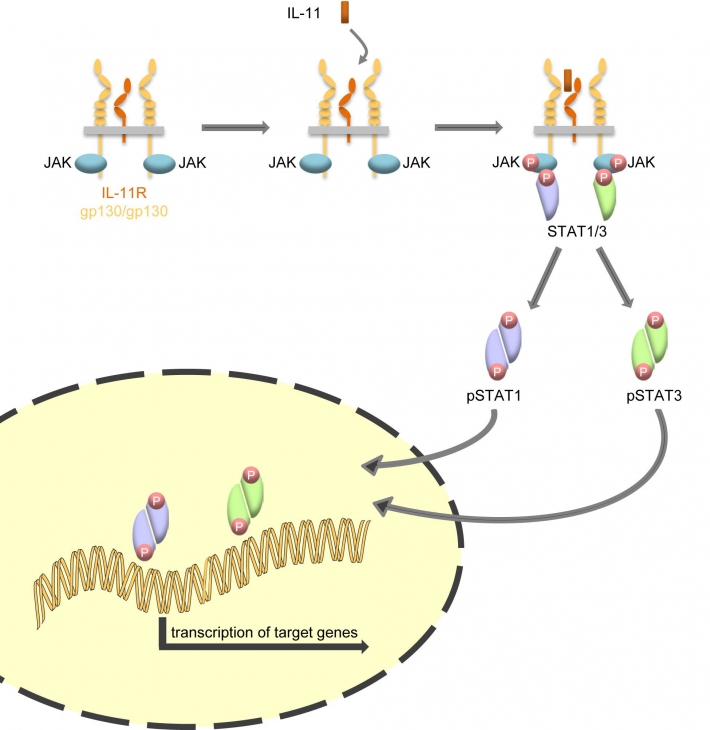
Figure 1
In healthy humans, IL-6 family cytokines are only found in very low concentrations or are even completely absent in the serum. However, levels can increase dramatically up to 100,000 fold, especially under inflammatory, pathophysiological conditions. To avoid uncontrolled cytokine signaling, which is itself associated with chronic inflammation, autoimmunity and cancer, cellular expression of cytokine receptors must therefore be tightly controlled and regulated.
Post-translational modifications have a huge impact on protein functions. We are particularly interested in the limited proteolysis (ectodomain shedding) of membrane-tethered cytokine receptors.
Signaling of IL-6 via the membrane-bound IL-6R is termed classic signaling, whereas signaling via the soluble IL-6R (sIL-6R) has been designated as trans signaling. As the example of the IL-6R illustrates, shedding through the two membrane-bound proteases ADAM10 and ADAM17 results in the generation of a soluble IL-6R (sIL-6R). The sIL-6R binds its ligand IL-6 with similar affinity as the IL-6R on the cell surface, and the IL-6/sIL-6R complex can activate virtually all cells of the human body, since the signal-transducing β-receptor gp130 is ubiquitously expressed. Thus, IL-6R shedding represents a way to generate an agonistic soluble cytokine receptor, which can in complex with its ligand afterwards activate cells that normally are not responsive to IL-6 (Figure 2).
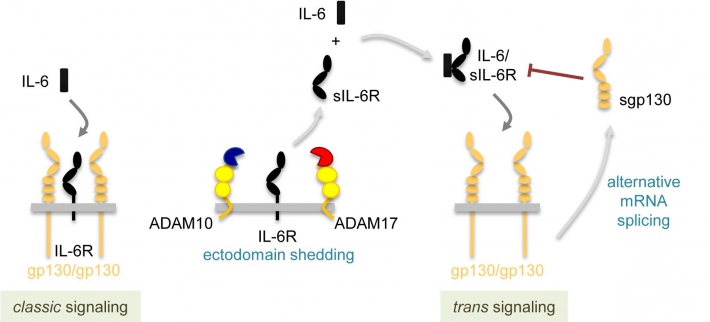
Figure 2
Besides this, ectodomain shedding represents a mechanism how cells can down-regulate their cytokine receptor expression, making them unresponsive towards stimulation by the cytokine themselves.
Funding
We are part of the Collaborative Research Center 877 (Proteolysis as a Regulatory Event in Pathophysiology) which is funded by the Deutsche Forschungsgemeinschaft (DFG).
Working with us
We always offer experimental bachelor and master thesis for students of natural sciences as well as experimental doctoral thesis for medical students. For inquiries and further information please contact Christoph Garbers.
| Contact: Prof. Dr. rer. nat. Christoph Garbers, phone 0391-67-15488 | christoph.garbers@med.ovgu.de |








